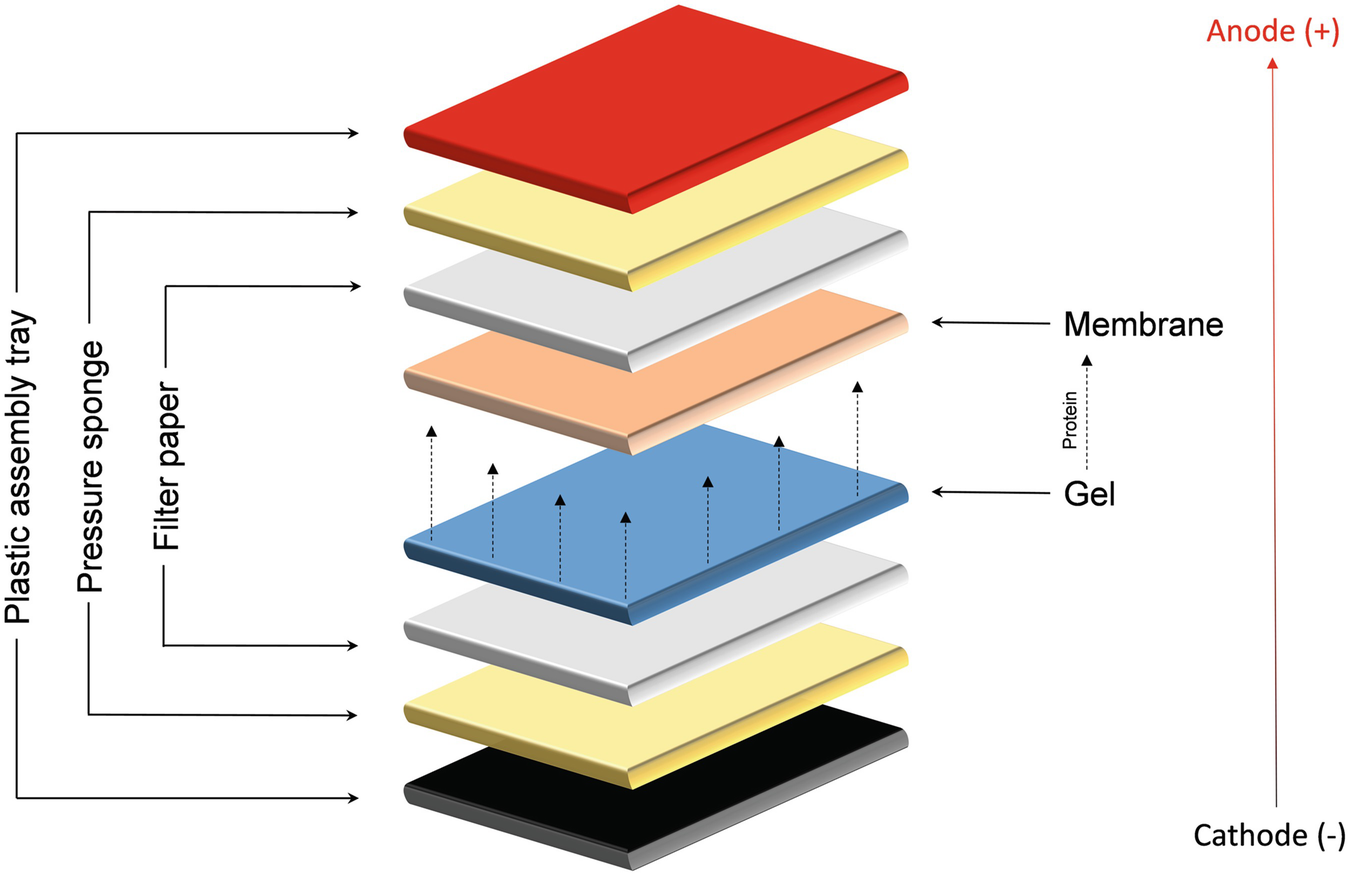
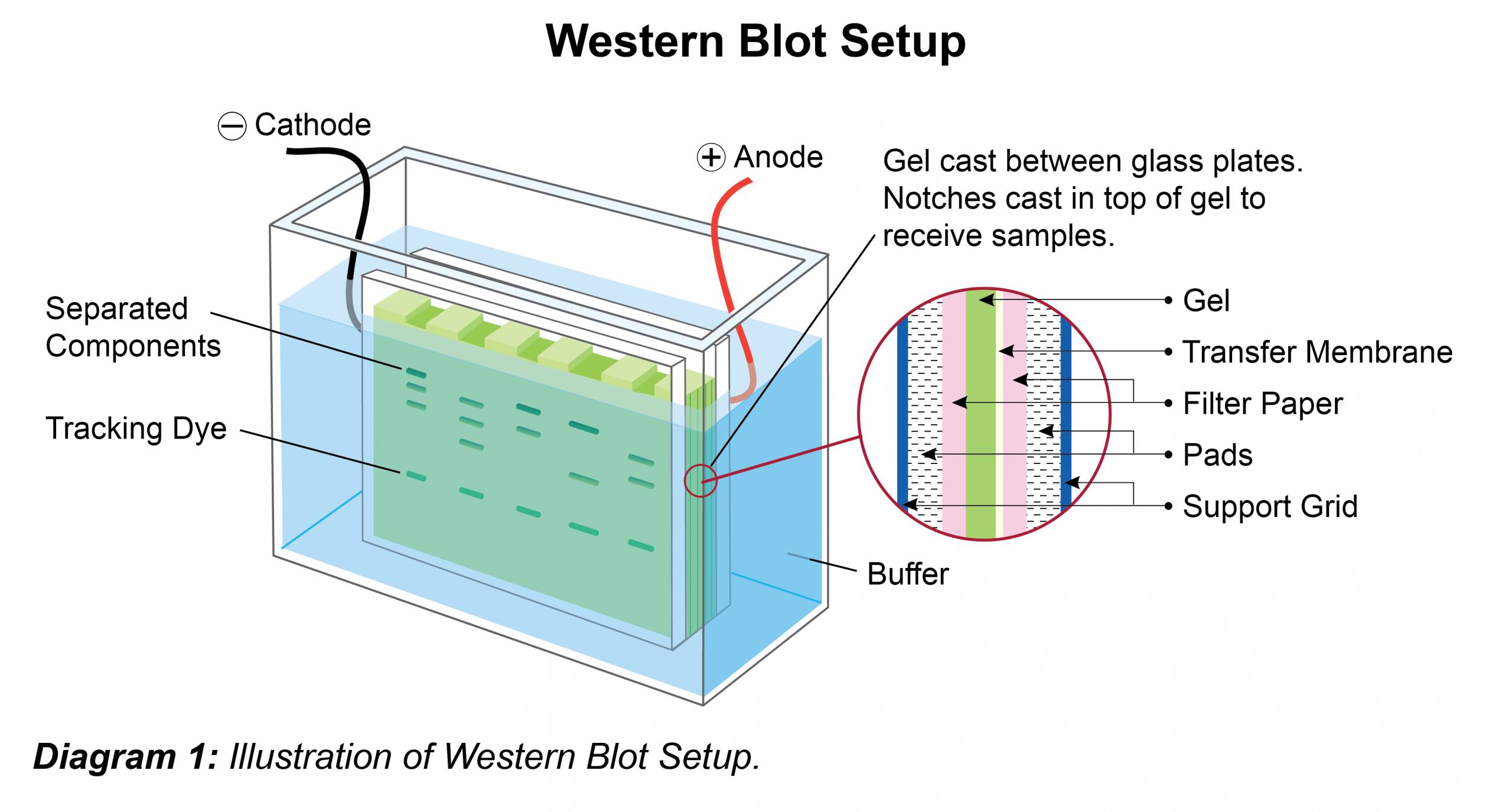
Western blotting was developed by George Stark’s group at Stanford University in 1979. Southern blotting was developed by Edward M. Southern blotting is used to find out a specific DNA sequence whereas, western blotting is used to find out a specific amino acid or protein sequence.ĬONTINUE READING BELOW Comparison Chart Southern BlottingĪ technique that is used to find out a specific DNA sequence in a given mixture is known as southern blotting.Ī technique that is used to find out a specific amino acids sequence of the protein in a given mixture is known as southern blotting. This name was used to match with southern blotting. On the other hand, western blotting was developed by George Stark’s group at Stanford University in 1979. Southern blotting is the type of blotting that is used to detect DNA while western blotting is used to find out protein. There are different types of blotting on the basis of detection of specific material, i.e., southern blotting, northern blotting, and western blotting. These molecules are pressed against the surface of an immobilized membrane which transfers the molecules onto the membrane. Here, minute particles will move faster than the bigger ones. In this technique, macromolecules like nucleic acids (DNA and RNA) and proteins in a mixture move through a slab of gel. Western Blottingīlotting is a technique that is used by scientists to separate different types of molecules from a mixture or sample. Transfer efficiencies of 60 to 80% may be achievable for proteins between 14 and 116 kDa, longer transfer times are required to transfer higher molecular weight proteins.The main difference between Southern Blotting and Western Blotting is that Southern Blotting is a technique that is used to detect a specific DNA fragment in a given sample whereas Western Blotting is used to find out a specific protein in a given sample. Methanol-free transfer buffers are recommended to reduce transfer time to 7 to 10 minutes. Electrotransfer is performed either at constant current (0.1 up to ~0.4 A) or voltage (10 to 25 V) for 10 to 60 minutes. Methanol may be included in the transfer buffer, but other organic solvents, including aromatic hydrocarbons, chlorinated hydrocarbons and acetone, should not be used to avoid damage to the semi-dry blotter. One to four gels may be rapidly electroblotted to membranes. Likewise, it is key that the filter paper sheets and membrane are cut to the size of the gel. To maximize the current passing through the gel instead of around the gel, the amount of buffer available during transfer is limited to that contained in the sandwich, so it is helpful if the extra-thick filter paper (~3 mm thickness) and membrane are also sufficiently soaked in buffer. For this semi-dry transfer, it is very important that the gel is pre-equilibrated in transfer buffer. With increasing time, however, there is a risk of over-transfer (blew through) of the proteins through the membrane, especially for lower molecular weight (<30 kDa) proteins when using membranes with a larger pore size (0.45 µm).įor semi-dry protein transfer, the transfer sandwich is placed horizontally between two plate electrodes in a semi-dry transfer apparatus. The transfer efficiency improves with increased transfer time. Transfer efficiencies of 80–100% are achievable for proteins between 14–116 kDa. A high field option exists for a single gel, which may bring transfer time down to as little as 30 minutes, but it requires the use of high voltage (up to 200 V) or high current (up to 1.6 A) and a cooling system to dissipate the tremendous heat produced. Transfers are typically performed with an ice pack and at 4☌ to mitigate the heat produced. Multiple gels may be electrotransferred in the standard field option, which is performed either at constant current (0.1 to 1 A) or voltage (5 to 30 V) from as little as 1 hour to overnight. The supported gel sandwich is placed vertically in a tank between stainless steel/platinum wire electrodes and filled with transfer buffer. The gel is then placed in the “transfer sandwich” (filter paper-gel-membrane-filter paper), cushioned by pads and pressed together by a support grid. When performing a wet transfer, the gel is first equilibrated in transfer buffer. SureLock Tandem Midi Gel Tank: capacity for up to 2 blot modules XCell SureLock Mini-Cell: capacity for up to 2 blot modules Mini Gel Tank: capacity for up to 2 Mini Blot Modules
Pre-assembled Select transfer stacks: buffer not required 1 mini gel per blot module 1–2 blot modules per tankġ midi gel per blot module 1–2 blot modules per tank
